Hydrogen Spectrum Emission Levels #1

by Carlos Clarivan
Title
Hydrogen Spectrum Emission Levels #1
Artist
Carlos Clarivan
Medium
Photograph - Photograph
Description
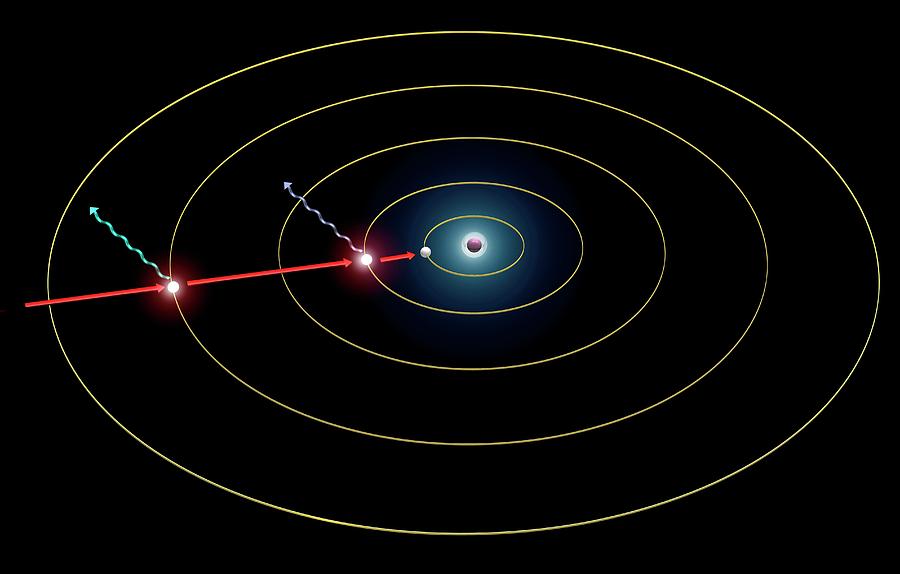
Hydrogen spectrum emission levels. Diagram showing the quantum basis for the emission spectrum of a hydrogen atom. When electrons in an element become excited (by heating), they enter higher energy orbits. When they return to their ground state they release the extra energy as light radiation at a specific wavelength characteristic of that element. Shown here (electrons in their excited states) are: the 4-2 transition, emitting blue-green light at 486.1 nanometres, part of the Balmer series; and the 2-1 transition, emitting ultraviolet light at 121.5 nanometres, part of the Lyman series. For this diagram with labels, see C025/8084.
Uploaded
July 29th, 2016
Embed
Share
Comments
There are no comments for Hydrogen Spectrum Emission Levels #1. Click here to post the first comment.






































