Hydrogen Bonding In Water

by Carlos Clarivan
Title
Hydrogen Bonding In Water
Artist
Carlos Clarivan
Medium
Photograph - Photograph
Description
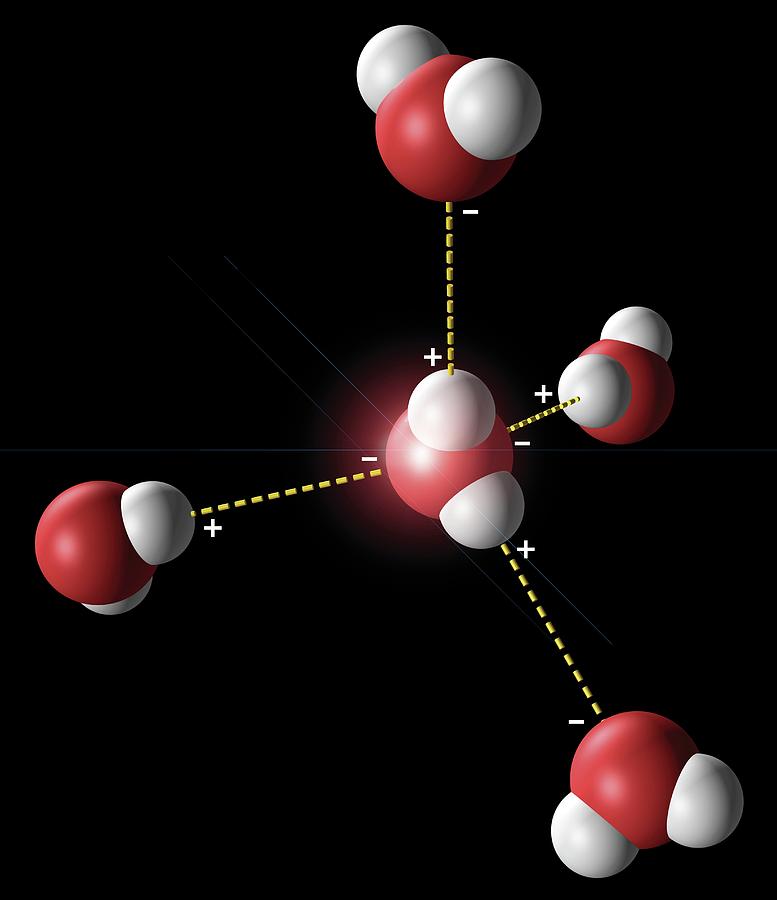
Hydrogen bonding in water. Artwork showing the hydrogen bonding (yellow dotted lines) between five water molecules. There is a weak, partial negative charge on the oxygen atoms (red), and a partial positive charge on the hydrogen atoms (white). Thus when water molecules are close together, their positive and negative regions are attracted. The force of attraction is called a hydrogen bond. The oxygen of one water molecule has two lone pairs of electrons, each of which can form a hydrogen bond with a hydrogen on another water molecule. This can repeat such that every water molecule is hydrogen-bonded with up to four other molecules.
Uploaded
June 30th, 2016
Embed
Share






































