Titanium

by Science Photo Library
Title
Titanium
Artist
Science Photo Library
Medium
Photograph - Photograph
Description
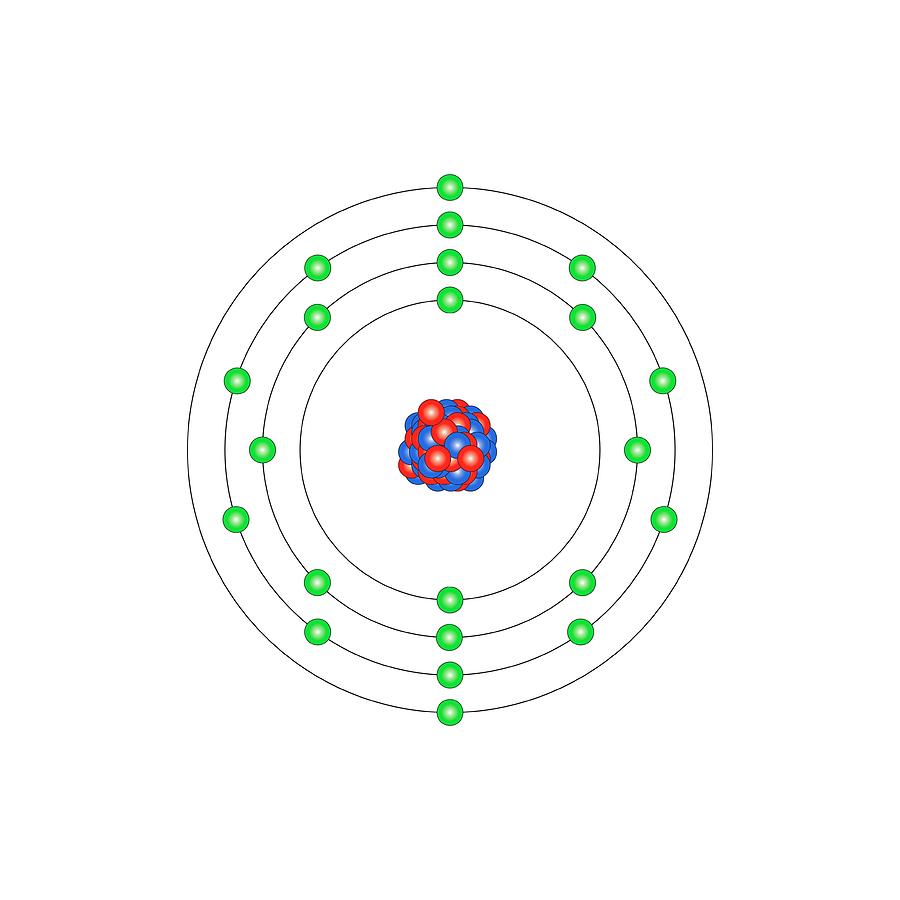
Titanium (Ti). Diagram of the nuclear composition and electron configuration of an atom of titanium-48 (atomic number: 22), the most common isotope of this element. The nucleus consists of 22 protons (red) and 26 neutrons (blue). 22 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Titanium is a transition metal in group 4, period 4, and the d-block of the periodic table. It has a melting point of 1668 degrees Celsius. The trends across the transition metals are due to electrons filling an inner d-subshell (here, within the 3rd ring), shielding the outer electrons from the increasing nuclear charge.
Uploaded
September 24th, 2018
Embed
Share
Comments
There are no comments for Titanium. Click here to post the first comment.






































