Sulfur

by Carlos Clarivan
Title
Sulfur
Artist
Carlos Clarivan
Medium
Photograph - Photograph
Description
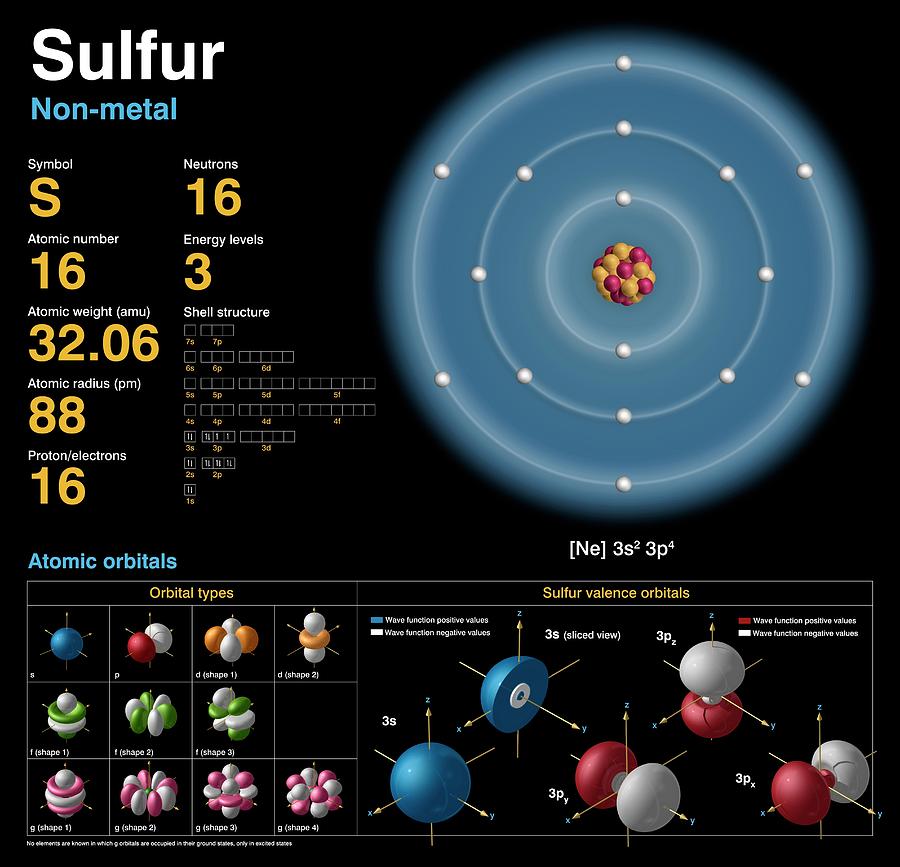
Sulfur (S). Diagram of the nuclear composition, electron configuration, chemical data, and valence orbitals of an atom of sulfur-32 (atomic number: 16), the most common isotope of this element. The nucleus consists of 16 protons (red) and 16 neutrons (orange). 16 electrons (white) occupy available electron shells (rings). The stability of an element's outer (valence) electrons determines its chemical and physical properties. Sulfur, a solid non-metal, is in group 16, period 3, and the p-block of the periodic table. Its compounds are vital for life. In elemental form there are numerous allotropes, the most common of which is octasulfur.
Uploaded
June 28th, 2016
Embed
Share







































