Limestone-acid Reaction

by Science Photo Library
Title
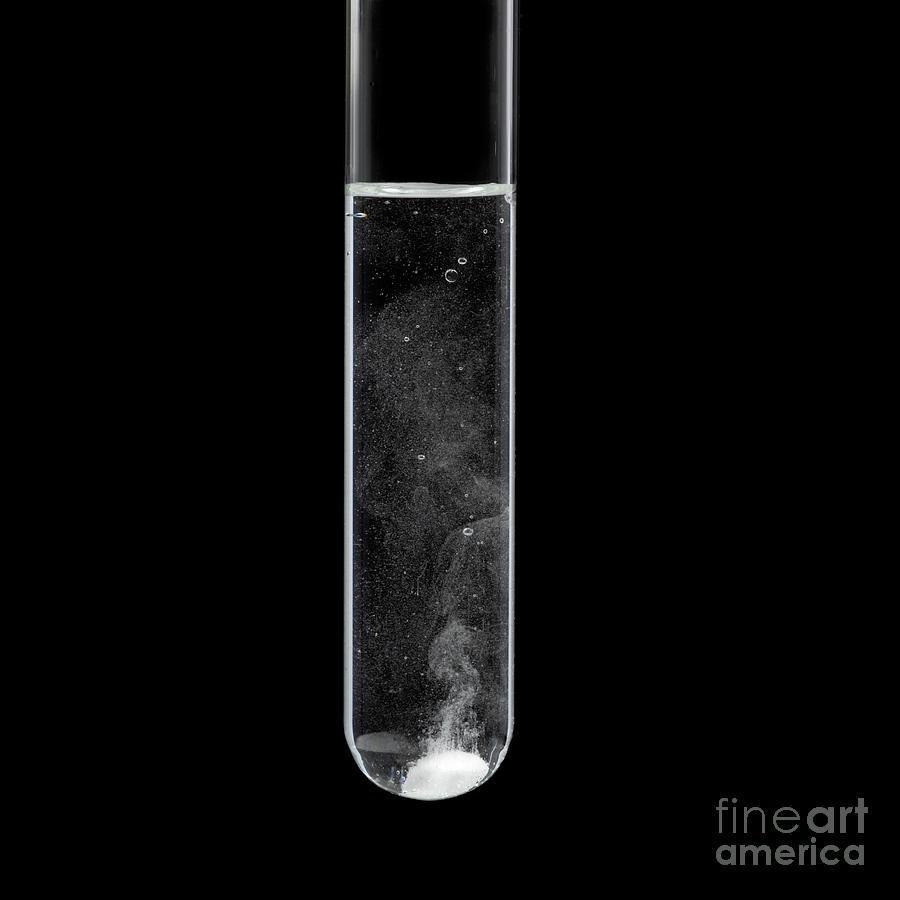
Limestone-acid Reaction
Artist
Science Photo Library
Medium
Photograph - Photograph
Description
Limestone-acid reaction. Limestone (calcium carbonate, CaCO3) reacting with acid in a test tube. Bubbles of carbon dioxide gas are being produced in the resulting reaction. This is an acid-base reaction, where calcium carbonate (the base) reacts with the acidic hydrogen (H+) to form aqueous calcium ions and carbonic acid (H2CO3). This acid is unstable, and rapidly decomposes to form carbon dioxide (CO2) and water (H2O). This is how rainwater erodes limestone.
Uploaded
September 25th, 2019
Statistics
Viewed 618 Times - Last Visitor from Ottawa, ON - Canada on 04/25/2024 at 9:41 AM
Embed
Share
Sales Sheet
Comments
There are no comments for Limestone-acid Reaction. Click here to post the first comment.





































