Electrolysis Of Water #4

by Mikkel Juul Jensen / Science Photo Library
Title
Electrolysis Of Water #4
Artist
Mikkel Juul Jensen / Science Photo Library
Medium
Photograph - Photograph
Description
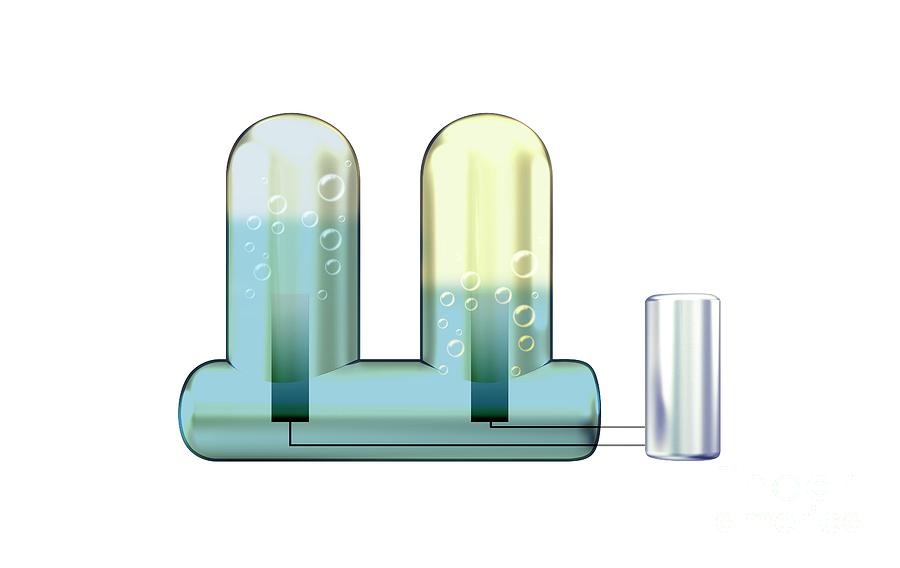
Electrolysis of water, illustration. Electrolysis is the use of an electrical current to decompose a chemical, in this case water. Reactions at the two electrodes (anode and cathode) are powered by the electric current from the battery (right). The electric current splits the water (H2O) into its elemental parts. Oxygen (O2) and hydrogen (H2) gas bubbles are evolved at the anode (left electrode) and cathode (right electrode) respectively. As water molecules consist of two hydrogen atoms and one oxygen atom, twice as much hydrogen as oxygen is trapped in the test tubes. First carried out in 1800, this is a key science experiment taught in schools. For this image with different background and with and without labels, see C046/8153 to C046/8159.
Uploaded
November 21st, 2019
Embed
Share
Comments
There are no comments for Electrolysis Of Water #4. Click here to post the first comment.






































