Structural Symmetry Of An Emerald

by Greg Williams/science Photo Library
Title
Structural Symmetry Of An Emerald
Artist
Greg Williams/science Photo Library
Medium
Photograph - Photograph
Description
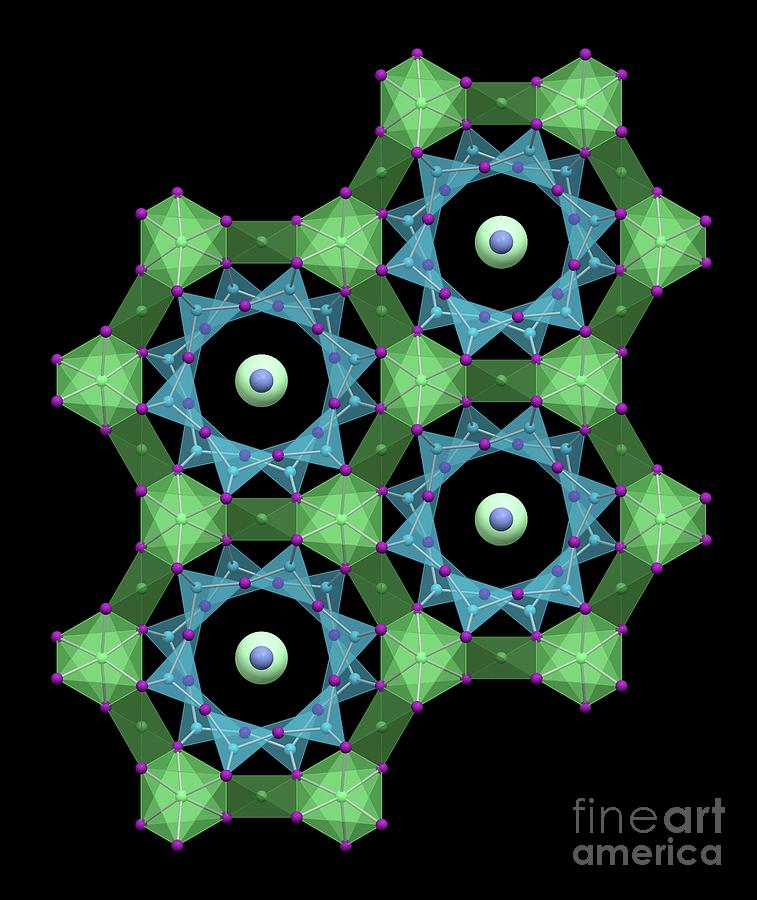
Structural symmetry of an emerald, illustration. Emerald is a variety of beryl, a mineral built around 12-membered crown-shaped rings of six tetrahedral silicon atoms (blue) alternating with six bridging oxygen atoms (violet). Each silicon atom is also bonded to two other oxygen atoms that are shared with beryllium(2+) and aluminium(3+) cations. These metal ions form a second ring that encloses the first. The outer ring has six aluminium oxide (AlO6) octahedra (light green) alternating with six tetrahedral beryllium oxide (BeO4) groups (darker green). The rings create channels that contain caesium(+) (yellow-green) and sodium(+) (lavender) ions. Pure beryl is colourless, but trace amounts of impurities give coloured gemstones. Emerald is a green form of beryl contaminated with chromium(3+) in some of the aluminium(3+) sites. The pale blue aquamarine has iron(2+) impurities; iron(3+) impurities give golden heliodor, and morganite is pink due to manganese(2+).
Uploaded
November 16th, 2019
Embed
Share
Comments
There are no comments for Structural Symmetry Of An Emerald. Click here to post the first comment.






































