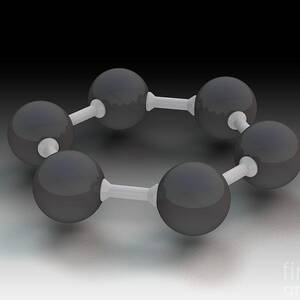
Graphite Molecular Structure #1

by Mikkel Juul Jensen/science Photo Library
Title
Graphite Molecular Structure #1
Artist
Mikkel Juul Jensen/science Photo Library
Medium
Photograph - Photograph
Description
Graphite molecular structure, illustration. Graphite is a form (allotrope) of the element carbon. Graphite is used in pencil leads and as a lubricant. It is composed of parallel layers of hexagonally arranged carbon atoms (spheres). Within each layer the carbon atoms are linked by strong covalent bonds, while the parallel layers are linked together by weak Van der Waals' forces. This Van der Waals' bonding is strong enough to hold the layers together, yet weak enough to let them slide over each other. This results in graphite's softness and its ability to act as a lubricant. For illustrations showing the hexagonal unit cell, see images C042/4538 to C042/4541.
Uploaded
November 8th, 2019
Embed
Share
Comments
There are no comments for Graphite Molecular Structure #1. Click here to post the first comment.