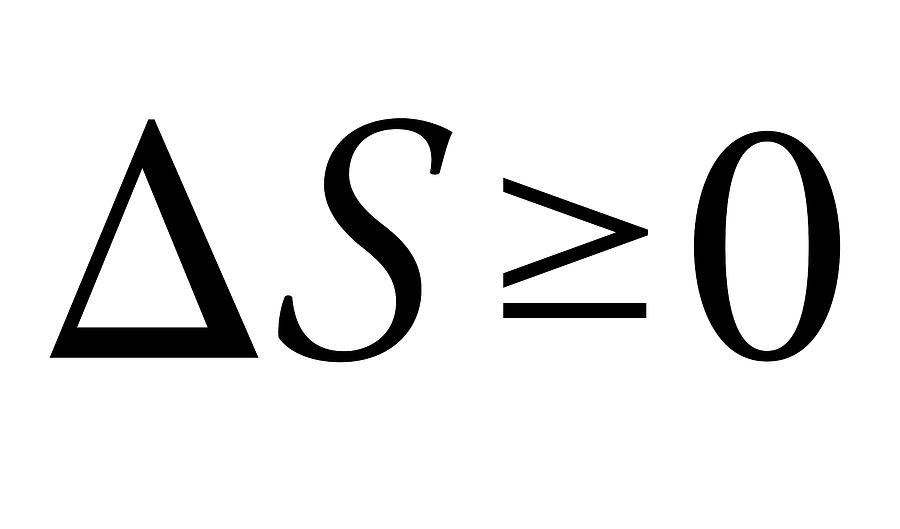
Second Law Of Thermodynamics

by Science Photo Library
Title
Second Law Of Thermodynamics
Artist
Science Photo Library
Medium
Photograph - Photograph
Description
Second law of thermodynamics, equation. This law states that the entropy (dS) of an isolated system not in equilibrium will tend to increase over time, approaching a maximum value at equilibrium. Entropy is a physics term to describe the amount of disorder in a system. When applied to the formula, it means that energy in the universe is gradually moving towards disorder. In simple terms, the second law of thermodynamics can describe how heat can flow from a hotter (high entropy) to a colder body (low entropy) but not the reverse. In 1824, the French physicist Sadi Carnot, first published these ideas by observing and measuring how steam engines operated.
Uploaded
July 31st, 2016
Statistics
Viewed 1,993 Times - Last Visitor from Ottawa, ON - Canada on 04/19/2024 at 12:01 PM
Embed
Share
Sales Sheet






































