Hydrogen Emission And Absorption Spectra #1

by Carlos Clarivan
Title
Hydrogen Emission And Absorption Spectra #1
Artist
Carlos Clarivan
Medium
Photograph - Photograph
Description
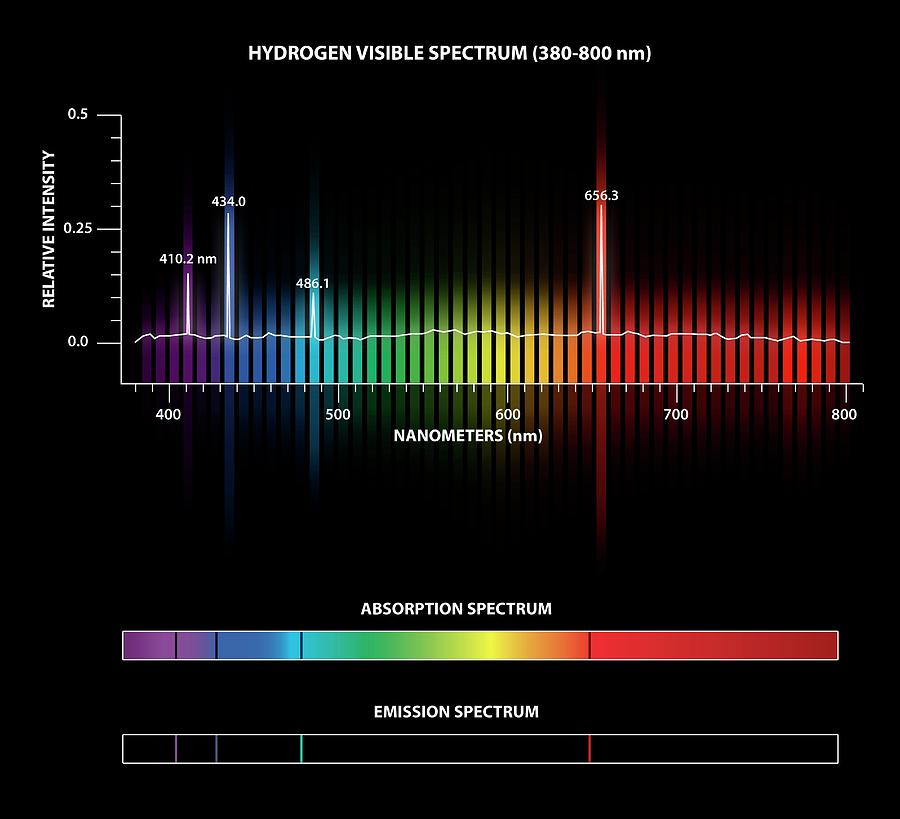
Hydrogen emission and absorption spectra. When electrons in an element become excited (by heating), they enter higher energy orbits. When they return to their ground state they release the extra energy as light radiation at a specific wavelength. The wavelengths emitted by an element are characteristic of that element. A matching absorption spectrum occurs when light passes through a material and is absorbed by its atoms. The 4 lines of the visible hydrogen spectrum (the Balmer series) correspond to wavelengths of 410.2, 434.0, 486.1 and 656.3 nanometres (nm). For this diagram with unlabelled lines, see C025/8083.
Uploaded
July 29th, 2016
Statistics
Viewed 1,230 Times - Last Visitor from Ottawa, ON - Canada on 04/18/2024 at 10:59 PM
Embed
Share
Sales Sheet







































