Chlorine #1

by Carlos Clarivan
Title
Chlorine #1
Artist
Carlos Clarivan
Medium
Photograph - Photograph
Description
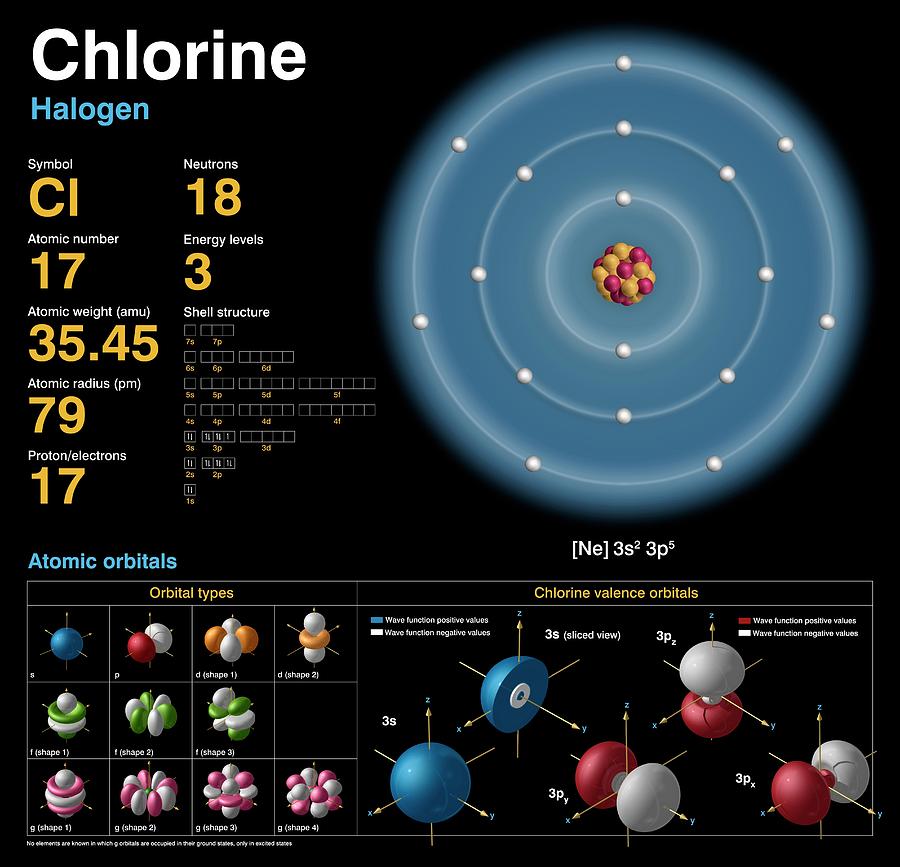
Chlorine (Cl). Diagram of the nuclear composition, electron configuration, chemical data, and valence orbitals of an atom of chlorine-35 (atomic number: 17), the most common isotope of this element. The nucleus consists of 17 protons (red) and 18 neutrons (orange). 17 electrons (white) occupy available electron shells (rings). The stability of an element's outer (valence) electrons determines its chemical and physical properties. Vital to life in ionic form, chlorine is a halogen in group 17, period 3, and the p-block of the periodic table. In elemental form it is a yellow-green, reactive, toxic gas (Cl2) that liquefies at minus 34 degrees Celsius.
Uploaded
August 3rd, 2016
Statistics
Viewed 1,327 Times - Last Visitor from Ottawa, ON - Canada on 04/19/2024 at 4:21 PM
Embed
Share
Sales Sheet
Comments
There are no comments for Chlorine #1. Click here to post the first comment.






































